Article Text
Abstract
Background The Canada-Denmark (CANDEN) definitions of spinal MRI lesions allow a detailed anatomy-based evaluation of inflammatory and structural lesions in vertebral bodies and posterior elements of the spine in patients with axial spondyloarthritis (axSpA). The objective was to examine the reliability, responsiveness and discrimination of scores for spinal inflammation, fat, bone erosion and new bone formation based on the CANDEN system and to describe patterns of inflammatory and structural lesions and their temporal development.
Methods 49 patients with axSpA from an investigator-initiated, randomised, placebo-controlled trial of adalimumab underwent spinal MRI at weeks 0/6/24/48. MR images were scored according to the CANDEN system and the Spondyloarthritis Research Consortium of Canada (SPARCC) method. Total scores, and various subscores, were created by summing individual lesion scores.
Results The CANDEN spine inflammation score had high responsiveness, similar to the SPARCC MRI spine index (Guyatt’s responsiveness index 1.88 and 1.67, respectively), and discriminated between adalimumab and placebo treatment already at 6 weeks’ follow-up (P=0.03). Anterior/posterior corner inflammation subscores showed similar responsiveness. Inter-reader reliability for the CANDEN spine inflammation and fat scores was good to very good for status and change scores (intraclass correlation coefficient (ICC)=0.71–0.92). Reliability for CANDEN new bone formation and erosion scores was good to very good for status scores (ICC=0.61–0.75) but, due to minimal progression, poor for change scores (ICC≤0.40).
Conclusions The CANDEN spine inflammation score showed good responsiveness, discrimination between active treatment and placebo and reliability. The CANDEN spine structural scores had good cross-sectional reliability, but longer studies are needed to investigate their sensitivity to change.
Trial registration number NCT01029847; Results.
- magnetic resonance imaging
- spondylarthritis
- outcomes research
- inflammation
This is an Open Access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
Statistics from Altmetric.com
Key messages
What is already known about this subject?
Patients with axial spondyloarthritis (axSpA) have different trajectories of inflammatory and structural spinal lesions during the disease course.
Detailed scoring of MR images of the vertebral bodies and posterior elements of the spine is possible using the 2009 Canada-Denmark (CANDEN) definitions for inflammatory and structural spinal lesions.
What does this study add?
The CANDEN spine inflammation score showed good reliability, responsiveness and discriminated well between active treatment and placebo, and it has inherent good face validity as posterior elements of the spine are also assessed.
How might this impact on clinical practice?
The CANDEN scores are not intended to be used to diagnose axSpA, but rather to allow detailed monitoring in research settings of changes over time in patients who have already been diagnosed with axSpA.
Introduction
Axial spondyloarthritis (axSpA) is a chronic inflammatory disease that affects the sacroiliac joints and the spine.1 MRI can detect inflammatory lesions (bone marrow oedema (BME)/osteitis), and structural lesions (erosions, bone spurs, ankylosis and fat metaplasia). The histopathological correlation of MRI inflammation and structural changes are mostly unknown, but BME of the facet joints has been shown to correlate with inflammatory cell infiltrates.2
In 2009, the founders of the Canada-Denmark (CANDEN) MRI working group (RL, WM, MØ, SJP) developed definitions and an atlas of inflammatory3 and structural lesions4 of the spine, and reported the reliability of scoring the individual lesions in a multireader exercise.5 6 The CANDEN approach allows a systematic assessment of MR images of the spine from the perspective of diagnostic ascertainment, and of quantifying both active inflammatory and structural abnormalities according to their precise anatomical locations and their relations over time.7 The definitions were based on sagittal T1-weighted and Short Tau Inversion Recover (STIR) MRI sequences of the vertebral bodies and the posterior elements of the vertebrae (ie, the costotransverse, costovertebral and facet joints, transverse and spinous processes and the adjacent soft tissues). Since then, a score for spinal fat lesions (Fat Spondyloarthritis Spine Score (FASSS))8 and a score for new bone formation,9–11 based on the CANDEN MRI definitions has been developed. Reference images are available online.12
The Spondyloarthritis Research Consortium of Canada (SPARCC) MRI spine index is a well-established MRI scoring method for spinal inflammation. The standard SPARCC 23-discovertebral unit (DVU) and 6-DVU methods cover the vertebral bodies,13 but not lesions located in the posterior elements of the spine or the soft tissues adjacent to the spine. An extended SPARCC scoring method that captured lesions in the posterior elements has been developed,14 but has not been used in clinical trials. The SPARCC method is applied on only three consecutive sagittal slices of each DVU and may therefore not provide information on the total inflammatory load of the patients. In contrast, the CANDEN MRI definitions for inflammatory and structural lesions comprise detailed information on the anatomical location of lesions in the vertebral bodies as well as in the posterior elements (figure 1), and may therefore provide more accurate information on the total inflammatory load and allow detailed analyses of changes in lesions at specific locations over time.
(A) User interface used for scoring of sagittal images according to the Canada-Denmark scoring system. Twenty-three discovertebral units (DVUs) are assessed. Fat lesions are scored in a similar way as inflammatory lesions, except that the posterior elements (FIL, SP, ST and TP/R) are not assessed for fat lesions. Fat lesions follow the principles of inflammation, except that posterior elements are not assessed. aCIL, anterior corner inflammatory lesion; NIL, non-corner inflammatory lesion; pCIL, posterior corner inflammatory lesion; FIL, facet joint inflammatory lesion; SP, spinous process inflammatory lesion; ST, soft tissue inflammatory lesion; aLIL, anterior lateral inflammatory lesion; pLIL, posterior lateral inflammatory lesion; TP/R, transverse process/rib; aCOBE, anterior corner bone erosion; pCOBE posterior corner bone erosion; NOBE, non-corner bone erosion; aCANK, anterior corner ankylosis; pCANK, posterior corner ankylosis; NANK, non-corner ankylosis; aCOS, anterior corner spur; pCOS, posterior corner spur; NOS, non-corner spur; FANK, facet joint ankylosis. (B) Axial view of inflammatory lesions. Note that as sagittal images are used for scoring, the boundaries illustrated in axial view may vary a few millimeters depending on the exact placement of the sagittal slices, when following the definition of central and lateral slices.
The aim of this study was to develop and validate an anatomy-based inflammation score, the CANDEN spine inflammation score and to further develop and validate CANDEN spine scores of erosion, fat and new bone formation. Moreover, we aimed to apply the CANDEN MRI definitions for spinal inflammation, fat metaplasia, erosion and new bone formation in an investigator-initiated, randomised, double-blind, placebo-controlled trial of adalimumab versus placebo to describe the frequencies of inflammatory and structural lesions and their development over time.
Methods
Study design
Forty-nine patients participated in a randomised, double-blind, placebo-controlled trial of adalimumab versus placebo (ASIM trial, ClinicalTrials.gov NCT01029847). All patients fulfilled the Assessment in Spondyloarthritis International Society (ASAS) criteria for axSpA and had sacroiliitis on MRI and/or radiography. The patients were randomly assigned to adalimumab 40 mg subcutaneously every other week or placebo for 6 weeks, thereafter all patients were treated with adalimumab 40 mg subcutaneously every other week to week 48. The six patients not responding at week 24 had their treatment changed to another tumour necrosis factor (TNF) inhibitor at the discretion of the treating rheumatologist. Further details of this trial, including whole-body MRI outcomes, are described in a separate article.15
The study took place at five rheumatology outpatient clinics in The Capital and Zealand Regions of Denmark from February 2010 to March 2014. All imaging examinations were performed at the Department of Radiology, Copenhagen University Hospital Herlev.
Image acquisition and scoring
MRI of the spine was performed at baseline and at weeks 6, 24 and 48. The spine was imaged in three parts (separately for cervical, thoracic and lumbar spine). MRI was performed in a single high-field Philips Medical Systems Achieva scanner (3.0 T) by sagittal T1-weighted turbo spin echo (repetition time: 518 ms, echo time: 8 ms, matrix (cerv./thor./lumb.): 200×143/300×213/300×213, field of view (FOV) (cerv./thor./lumb.): 178×150 mm/270×150 mm/270×150 mm, slice thickness 4 mm, interslice gap 0.4 mm) and STIR (repetition time: 4990–8530 ms, echo time: 80 ms and inversion time: 120 ms, matrix (cerv./thor./lumb.): 272×212/272×215/272×215, FOV (cerv./thor./lumb.): 270×230 mm/270×150 mm/270×230 mm, slice thickness 4 mm, interslice gap 0.8 mm).
All MRI scans were read in chronological order by an experienced (SJP) and a newly trained (SK) reader blinded to other imaging and clinical data. Images were scored according to the definitions in the CANDEN MRI spine system3–6 and by the 23-DVU SPARCC MRI spine index using a web-based data entry interface (COPECARE SAMRI (Spondyloarthritis MRI module), figure 1). To ensure proper calibration of the two MRI readers for the SPARCC method, one of the developers of the SPARCC system (WM) also scored all images for the 23-DVU SPARCC MRI spine index.
Radiographs were obtained at baseline and at 48 weeks and were scored in known chronology by the modified Stoke Ankylosing Spondylitis Spine Score (mSASSS) by one experienced reader (SJP) in known chronological order, blinded to all other data.
Scoring of inflammatory lesions
Inflammation, new bone formation and erosion scores were developed from the CANDEN definitions of lesions and scoring,5 6 in a similar way as FASSS. The development and validation of FASSS based on the CANDEN MRI definitions has previously been reported.8
The spine is divided into 23 levels from C2/C3 to L5/S1. Central sagittal slices are the sagittal slices that include the spinal canal; the pedicle may be partially seen but is not continuous between the vertebral body and posterior elements3 5 (figure 1). Lateral sagittal slices are the sagittal slices that are located lateral to the spinal canal, and either the pedicle must be continuous between vertebral body and posterior elements or the slice is lateral to the pedicle. Only BME is scored, except for posterior element soft tissue oedema. To calculate the CANDEN spine inflammation score, a set of scoring rules are defined.
Vertebral bodies
In central sagittal slices, anterior and posterior corner inflammatory lesions are scored as 0 (absent) or 1 (present); in DVUs in thoracic and lumbar spine (T1/T2 and below), a score of 1 is added for large lesions (≥25% of the anteroposterior (AP) diameter of the vertebral endplate and/or height of the vertebral body, perpendicular to the endplate).3 Non-corner inflammatory lesions (BME located between the corners of a vertebral body) are scored as 0 (absent), 2 (present); in DVUs in the thoracic and lumbar spine, a score of 2 is added for large non-corner lesions (≥25% of the height of the vertebral body, perpendicular to the endplate). It was decided to assign the score 2 for non-corner lesions, while 1 for corner lesions (both doubled if large, see definitions), because the area of this anatomical region is typically larger than the corner areas, and because each vertebral endplate contains two corner regions but only one non-corner region, thereby giving the same total weight to the two lesion types in the central sagittal slices. If a corner lesion in any central slice involves >50% of the AP diameter of the vertebra, it is counted as a combined corner and non-corner lesion.
In lateral sagittal slices in the thoracic and lumbar spine, BME is scored if located at the anterior or posterior corner; these antero-lateral and postero-lateral inflammatory lesions are scored as 0 (absent) or 1 (present). All lesions are scored based on anatomical location; for example, if a lesion is visible at the anterior corner on a central sagitttal slice and in the anterior half of the vertebral body on a lateral slice, it is scored as the combination of an anterior corner lesion (sagittal slice) and an antero-lateral corner lesion (lateral slice).
Posterior elements
For each of the 23 levels from C2/C3 to L5/S1: facet joint inflammatory lesions on left and right side, spinous process inflammatory lesions and soft tissue inflammatory lesions (hyperintense signal on STIR at ligaments and entheses at the posterior elements of the vertebrae) are each scored as 0 (absent) or 1 (present). For each of the 17 vertebrae from T1 to L5: rib/transverse process inflammatory lesions are scored as 0 (absent) or 1 (present) on left and right side. Inflammation in the costovertebral joints was not scored separately but as inflammation of rib/transverse process and/or postero-lateral corner inflammation, when present at these locations.
The total scoring range for the CANDEN spine inflammation score is 0–582. The range for the vertebral body subscore is 0–456, and the range for the posterior elements subscore is 0–126.
Scoring of structural lesions
Fat metaplasia (FASSS) is scored as previously described,6 8 the scoring range is 0–456. The method for scoring erosions is similar to the method for scoring spine inflammation, except that erosions are only assessed in central sagittal slices. Anterior and posterior corner erosions are scored 1, non-corner erosion is scored 2, in DVUs in the thoracic and lumbar spine, a score of 1 is added for large corner lesions and a score of 2 is added for large non-corner lesions. Erosions are not scored in the facet joints or other posterior elements. The scoring range for the CANDEN spine erosion score is 0–320. To obtain the CANDEN spine new bone formation score, at each level, the presence of anterior corner ankylosis, posterior corner ankylosis and non-corner ankylosis are each scored as 0 (absent) or 6 (present), anterior corner, posterior corner and non-corner bone spurs are each scored as 0 (absent) or 2 (present), facet joint ankylosis is scored as 0 (absent) or 1 (present), and all individual lesion scores are summed. The new bone formation scores were chosen to be consistent with the scores used in the radiographic mSASSS system that assigns a score of 2 for bone spur and a score of 3 for ankylosis (in total a score of 6 for two ankylosed corners that are opposing each other). Scoring range for the CANDEN spine new bone formation score is 0–460; the vertebral body subscore has a range of 0–414 and the facet joints subscore has a range of 0–46.
The estimated reading time including data entry for obtaining the CANDEN spine inflammation score was not recorded systematically, but judged to be in the range of 10–45 min for all four timepoints depending on the amount of inflammation, similar to the SPARCC method in most cases. Total scoring time for inflammation and structural lesions in the spine were in the range 15–90 min per patient for four timepoints.
Statistical analysis
Data were characterised by descriptive statistics and correlations were assessed by Spearman’s correlation analysis. Difference between groups in change from baseline to weeks 6, 24 and 48 was assessed by Mann-Whitney U tests with continuity correction according to the predefined statistical analysis plan. Analysis of covariance (ANCOVA) with group allocation and baseline score as covariates and changes in continuous outcome variables as dependent outcomes were performed as post hoc secondary analyses to take differences in baseline values into account. Standardised response mean (SRM) was calculated as the overall mean change score divided by the SD of the change scores, while Guyatt’s responsiveness index (GRI) was calculated as the mean change score in the adalimumab group divided by the SD of the change scores in the placebo group at week 6; values ≥0.8 were judged to represent a large degree of responsiveness.16 Inter-reader agreement was assessed using a two-way random effects single measure model of intraclass correlation coefficient (ICC)1 3 based on absolute agreement. ICCs≥0.6 were considered to represent good reliability, and ≥0.8 very good reliability. Smallest detectable change (SDC) was calculated from baseline to week 24 as 1.96×SDdifference in change scores/(√2√k) for the k=2 readers (SJP and SK).
All MRI results at the patient level are reported as the mean score of the two primary MRI readers (SJP and SK). All MRI results at the lesion level are reported for the most experienced primary reader (SJP) for conciseness; largely similar results were obtained for the other reader (SK, data not shown). No imputation of missing data was performed, 39 of 49 patients had complete data at week 48. All statistical analyses were performed using R V.3.4.0.
Results
The patients had mean age 38 years (range 20–61), 51% were males, 73% were HLA-B27 positive, mean symptom duration 12 years (range 0–45) and mean baseline Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) 6.4 (range 4.2–9.8). Twenty-eight patients (57%) fulfilled the modified New York radiographic criteria for ankylosing spondylitis.
The anatomical distribution of inflammatory lesions at baseline and changes during the study
At baseline, 40 patients (82%) had a CANDEN spine inflammation vertebral body subscore ≥1. Six patients (12%) had a CANDEN spine inflammation posterior elements subscore ≥1. Patients with inflammatory lesions in the posterior elements all had concomitant inflammatory lesions in the vertebral bodies, not necessarily at the same level. Overall, inflammatory lesions were seen at 161 (14%) of 1127 levels (table 1). Fourteen (9%) of the involved levels had inflammatory lesions of both vertebral bodies and posterior elements, 134 levels (83%) had inflammatory lesions of only the vertebral bodies and 13 levels (8%) had inflammatory lesions of only the posterior elements. Inflammatory lesions were most frequently located at the anterior, posterior, antero-lateral and postero-lateral corners of the thoracic or lumbar spine (figure 2). Facet joint inflammation comprised 26 (81%) of 32 inflammatory lesions in the posterior elements.
The anatomical distribution of inflammatory and structural lesions in the spine
The distribution at baseline of inflammatory and structural lesions across vertebral bodies and posterior elements at each level from C2/C3 to L5/S1. Bars represent the number of patients with lesion by type and anatomical location. BME, bone marrow oedema; ERO, erosion; FAT, fat lesion; NBF, new bone formation (ankylosis/bone spur); PE, posterior elements; VB, vertebral body.
Corner inflammatory lesions of the vertebral bodies were seen in 131 (16%) of 833 thoracic or lumbar DVUs. Fifty-three of these DVUs (40%) had lesions visible on both central and lateral slices, while 60 DVUs (46%) had lesions that were only visualised on central slices and 18 DVUs (14%) had lesions that were only visualised on lateral slices.
Most inflammatory lesions present at baseline had disappeared after 24 weeks, while new inflammatory lesions developed very infrequently (table 2).
The disappearance of inflammatory lesions of the spine between baseline and week 24 during adalimumab therapy and the development of new lesions by anatomical location
CANDEN spine inflammation score: discrimination
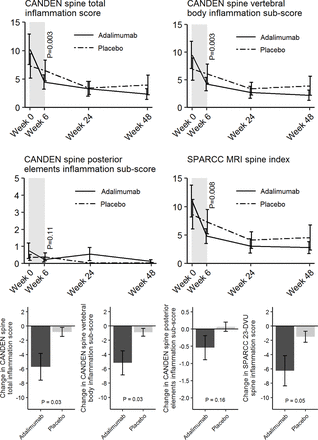
The mean CANDEN spine inflammation score was 8.8 (SD 12) at baseline. The vertebral bodies subscore had a mean value of 8.2 (SD 11) and constituted the major part of the total score. The posterior elements subscore had mean value 0.6 (SD 1.7). The CANDEN spine inflammation score decreased significantly more in the adalimumab group than in the placebo group at week 6 (table 3, figure 3). At weeks 24 and 48, no significant differences between groups remained (see online supplementary table 1). In post hoc secondary analyses, similar results were found using ANCOVA with baseline score as covariate, except that the difference between groups in SPARCC MRI spine index (23-DVU) at week 6 was highly significant when using ANCOVA (P=0.008) (figure 3). SPARCC results by the SPARCC developer can be found in online supplementary table 2.
Supplementary file 1
Construct validity as assessed by change in scores from baseline to week 6 in adalimumab group compared with placebo group
Mean values of CANDEN spine inflammation score and for vertebral body and posterior elements subscores, bars represent the SEM; as observed (n=49, 47, 42, 39). For comparison, change in SPARCC MRI spine index (23-DVU) is also shown. P values for difference between group at week 6 were calculated in post hoc secondary analyses by ANCOVA. Bar plots show changes from baseline to week 6 with P values for the difference between change values at week 6 calculated by Mann-Whitney U tests with continuity correction (per protocol); as observed (n=47). MRI scores were the average of the two readers. CANDEN,Canada-Denmark; DVU, discovertebral unit; SPARCC, Spondyloarthritis Research Consortium of Canada.
CANDEN spine inflammation score: responsiveness
The CANDEN spine inflammation score demonstrated a large degree of responsiveness at week 6 (GRI 1.88) comparable to the responsiveness of the SPARCC MRI spine index (GRI 1.67). The vertebral body subscore had a higher responsiveness (GRI 1.99) than the posterior elements subscore (GRI 0.84). At week 6, BASDAI (GRI 1.32) was less responsive than the CANDEN spine inflammation score and the SPARCC MRI spine index (table 4). At weeks 24 and 48, the responsiveness of the CANDEN spine inflammation score was moderate (SRM 0.57 and 0.55) and comparable with the SPARCC MRI spine index (SRM 0.57 and 0.55).
Baseline distribution and responsiveness of Canada-Denmark spine inflammation and structural scores compared with SPARCC and BASDAI
CANDEN spine inflammation score: construct validity
The CANDEN spine inflammation score was highly correlated with SPARCC MRI Spine Inflammation Index at baseline (rho=0.98, P<0.001), and changes in these two MRI measures were highly correlated (from week 0 to 6: rho=0.90, P<0.001; from week 0 to 24: rho=0.96, P<0.001). The CANDEN inflammation score correlated weakly with C-reactive protein (CRP) (rho=0.21, P=0.14), with the posterior elements subscore reaching statistical significance (rho=0.37, P=0.008), while the vertebral body subscore did not (rho=0.19, P=0.20). This was similar in magnitude to the correlation between SPARCC MRI Spine Inflammation Index and CRP (rho=0.18, P=0.22).
No significant correlations were found at baseline between CANDEN spine inflammation score and visual analogue scale (VAS) pain (rho=−0.13, P=0.37), BASDAI (rho=−0.11, P=0.45), Bath Ankylosing Spondylitis Functional Index (BASFI) (rho=−0.06, P=0.70) or Ankylosing Spondylitis Disease Activity Score (ASDAS) (rho=0.14, P=0.35). Changes in CANDEN spine inflammation score were not significantly correlated with changes in these clinical measures of disease activity and in CRP at week 6 or week 24 (data not shown).
The anatomical distribution of fat metaplasia at baseline and changes during the study
The most frequent structural lesions at baseline were fat anterior corner lesions in the thoracic and lumbar spine (figure 2). At the patient level, a decrease in CANDEN spine inflammatory score from baseline to week 24 was associated with an increase in the FASSS score (rho=−0.59, P<0.001). The mean increase in FASSS at week 24 was 1.7 (range −4 to 24). Change in FASSS at week 6 tended to be higher in the adalimumab group than the placebo group (table 3).
Twenty-four (3%) of 906 DVUs had an increase in fat score at week 24, 21 (88%) of these had a concomitant decrease in BME score, while 3 (13%) had no concomitant decrease in BME. Eighty-three DVUs had a decrease in BME without a concomitant increase in fat score at week 24. A decrease in fat at week 24 was observed in 5 DVUs. At the individual lesion level, 7 (3%) of 233 fat lesions present at baseline had disappeared at week 24, while 51 new fat lesions had developed, 43 (84%) of the new fat lesions were located in exactly the same locations where inflammation had been present at baseline.
Structural scores: responsiveness, construct validity and comparison with radiography
FASSS showed large responsiveness (GRI 0.41) at week 6, but low responsiveness (SRM 0.28 and 0.27) at weeks 24 and 48. Responsiveness for the CANDEN spine erosion score and the CANDEN spine new bone formation score was low during 48 weeks of follow-up, similar to mSASSS of radiographs (table 4). The CANDEN spine new bone formation score correlated with clinical examination (Bath Ankylosing Spondylitis Metrology Index (BASMI), rho=0.41, P=0.003) as well as with radiographic scores (mSASSS, rho=0.49, P<0.001). No significant correlations of the CANDEN spine erosion score were found.
MRI demonstrated new bone formation in nine (18%) patients at baseline (from C2/C3 to L5/S1). Ankylosis was found in eight (16%) patients; five patients had ankylosis of the vertebral bodies and six patients had ankylosis of the facet joints. In comparison, lateral radiographs showed new bone formation in 26 (53%) patients at baseline (from C2/C3 to C7/T1 and from T12/L1 to L5/S1); 9 patients had ankylosis of the vertebral bodies. Thus, MRI was markedly less sensitive in the lumbar and cervical areas than radiography, while the specificity for any spur/ankylosis by MRI was 99.6%, with radiographs as reference standard. Erosions and new bone formation remained largely unchanged as assessed by MRI. In comparison, two new bone spurs and ankylosis at three DVUs developed between baseline and week 48 as judged by radiographic scoring.
Inter-reader reliability and smallest detectable difference
CANDEN spine inflammation score had good to very good reliability for both status scores (inter-reader ICC of the 4 timepoints mean 0.82) and change scores (from baseline to the 3 follow-up timepoints mean 0.72), similar to that of the SPARCC MRI spine index (ICC 0.82 for status score, 0.76 for change scores). Inter-reader reliability of SPARCC for reader pairs WM-SJP/WM-SK was 0.80/0.79 for status scores and 0.86/0.83 for change scores, which confirms that the two CANDEN readers of this study (SJP and SK) were well calibrated according to the SPARCC method. The FASSS had a very good reliability (ICC mean 0.92 for status score and 0.71 for change score, respectively). Reliability for status score was good to very good for CANDEN spine erosion score (mean 0.61) and CANDEN spine new bone formation score (mean 0.75), while reliability for change scores were poor (all <0.40). At week 24, SDC was 7 points for the CANDEN total spine inflammation score; in comparison SPARCC score had a SDC of 8 points; six patients had an improvement in inflammation ≥SDC using either scoring method. The FASSS had a SDC of 2 points; at week 24, 12 patients had an increase above this threshold. Only limited changes in erosion score and new bone formation score were observed.
Discussion
We have developed and preliminarily validated a new, comprehensive set of anatomy-based MRI outcome measures for spinal inflammation (CANDEN spine inflammation score) and structural changes (CANDEN spine new bone formation score, CANDEN spine erosion score) in patients with axSpA in a randomised controlled trial based on previously published CANDEN MRI definitions.
The inflammatory component, the CANDEN spine inflammation score, possessed face, content and construct validity as well as good sensitivity to change and discrimination. Inflammation in axSpA involves different tissues, such as the fibrocartilaginous joints between the vertebral bodies that include the ring apophysis where the annulus fibrosus inserts, the synovial facet joints and costotransverse joints and the complex synovial costovertebral joints where most ribs articulate with facets on the margins of adjacent thoracic vertebral bodies and the intervertebral disc, as well as numerous entheseal sites, especially in the posterior elements. These different sites may respond differently to drugs with various modes of action. Thus, the detailed CANDEN system may allow an increased understanding of how different drugs may modify different aspects of the spinal disease.
In contrast to currently used MRI scoring methods of the spine such as the SPARCC system, which is based on assessment of the vertebral bodies on three consecutive slices at each discovertebral unit, and does not record whether inflammation is present as non-corner lesions, lateral lesions or anterior corner lesions, this anatomy-based assessment system provides this information. The system measures inflammation in the entire spine, including different parts of the posterior elements of the spine, providing greater face and content validity. First, the posterior elements constitute a large volume of the vertebrae and have large surface areas with insertions of ligaments and tendons, and there is no reason why inflammation here should be less clinically relevant to the patients. Second, the specificity of inflammation of the costotransverse, costovertebral and facet joints may be greater for axSpA, compared with the corner and particularly the non-corner inflammatory lesions of the vertebral bodies, which may be seen in healthy controls and in patients with degenerative disc disease.17–19
The pattern and prevalence of inflammatory and structural changes at different anatomical locations in a population of patients starting TNF inhibitor treatment was described, and found to be highest in the lower thoracic and lumbar spine. The frequency of posterior findings was lower than expected, as only six (12%) patients had inflammatory lesions of the posterior elements in this trial. Lesions in the posterior elements only gave a minor contribution to the total mean scores. Interestingly, all six patients with inflammatory lesions of the posterior elements were HLA-B27 positive and fulfilled the radiographical criteria for ankylosing spondylitis (AS), and 4 (67%) were clinical responders at week 24. This may suggest that posterior elements are rarely involved in nr-axSpA, but this needs further investigation.
Other studies have reported higher prevalences of involvement of the posterior elements. Bochkova et al found inflammatory lesions of the posterior elements in 22 (76%) of 29 patients that all fulfilled the radiographic criteria for AS and almost all (28 (97%) of 29) were HLA-B27 positive.20 They performed additional axial images at painful regions of the spine in case of normal sagittal images, which may increase sensitivity. Maksymowych et al found inflammatory lesions of the posterior elements in 28 (88%) of 32 patients with AS.14 Althoff et al found inflammatory lesions of the posterior elements more often in the AS group (10 (26%) of 39) than in the non-radiographic axSpA group (3 (8%) of 36).21 Larbi et al found inflammatory lesions in 23 of 112 (21%) of recently diagnosed HLA-B27-positive patients with SpA.22 In comparison, in our study 36 (73%) were HLA-B27 positive and 28 (57%) fulfilled the radiographical criteria for AS. Thus, the lower prevalence of posterior inflammatory lesions in our study may be due to a different case mix of patients compared with the other studies.
Most vertebral non-corner inflammatory lesions that were present at baseline had disappeared after 24 weeks (56%), and the number of new similar lesions that developed during the trial was minimal. Still, other inflammatory lesions such as anterior and posterior corner lesions disappeared even more frequently during treatment. Non-corner lesions are often seen as a part of endplate lesions similar to a Modic type I lesion, and disc degeneration rather than spondyloarthritis will be a probable cause of some of these lesions.
Rennie et al have reported that lesions in 20% of inflamed vertebral bodies were only visible on ‘lateral’ sagittal MRI slices.23 A comparable result was reached in our study, where 14% of inflamed vertebral bodies were only visualised on the lateral slices. The spatial distribution of syndesmophytes along the vertebral rim has been demonstrated by CT to involve most of the circumference, preferentially the postero-lateral rim,23 and including as much of the rim as possible in the scoring should increase the sensitivity of the instrument. This underlines the need for assessing central sagittal slices, and lateral sagittal slices, if a comprehensive score for all inflammatory lesions in the vertebral bodies is needed. The SPARCC spine method does assess lateral inflammation in the vertebral body, as the three consecutive slices approach can be applied to lesions in the lateral slices in the same manner as for central slices.
At baseline, only six patients (12%) had inflammatory lesions in the posterior elements, and all these had also lesions in the vertebral bodies. The GRI for the total CANDEN spine inflammation score and for the vertebral body subscore were almost identical. The addition of posterior elements data to the vertebral body subscore did not improve responsiveness. The low frequency of posterior lesions in this study likely accounts for the lack of impact of including these elements. The responsiveness measures SRM and GRI assume that change scores are normally distributed; many patients in this study had little spinal involvement and therefore no change over time, and these responsiveness measures would likely have been higher if the case mix had consisted of more patients with substantial spinal involvement. Importantly, the responsiveness of the CANDEN spine inflammation score was similar to that of the SPARCC MRI spine index. It is important to note that a method that focuses on assessing presence/absence and size of individual lesions, such as CANDEN, may perform differently than a method that has been designed to assess the total amount of inflammation, independent of location, such as SPARCC, in groups of patients with many spinal lesions as compared to those with few. Consequently, comparison of responsiveness and discrimination will be required in larger cohorts with a broader range of spinal lesions.
In this study, the fat metaplasia score (FASSS) tended to increase over time, and week 24 change in FASSS correlated with the disappearance of inflammatory lesions. This temporal association, and the fact that 84% of new fat lesions occurred in areas in which inflammation had been present at baseline, further validates the FASSS score as a measure of structural changes caused by inflammation. Bone spurs and ankylosis of the vertebral bodies were observed more frequently by radiography than MRI, but MRI allowed an assessment of the thoracic spine and facet joints that are difficult to delineate on lateral radiographs and that are not included in the mSASSS score for radiographic changes. Further studies with longer follow-up are needed to clarify the clinical importance of these structural measures as well as finding possible ways to improve their reliable detection by MRI.
It is important to note that the CANDEN scores are not intended to be used to diagnose axSpA, but rather to allow detailed monitoring of changes over time in patients who have already been diagnosed with axSpA. The value of the CANDEN system is that it allows more detailed analyses of the link between inflammation and structural changes and their development over time and at the same time may provide a patient-level score for spinal inflammation. Ten patients did not complete the trial,15 but since their baseline characteristics were similar to the other patients (data not shown), the results presented here were likely not impacted.
In conclusion, the present study documents the construct validity, responsiveness, discriminatory ability and reliability of the CANDEN spine inflammation score. The method is a promising outcome measure in patients with axSpA, as it both allows assessment of the total spinal inflammation, and also separate analyses of various articular, entheseal and discovertebral components of the disease. Through comprehensive assessment of all parts of the spine, it has inherently good face and content validity. The structural CANDEN spine scores showed good cross-sectional reliability, but further studies with longer follow-up are needed to clarify the validity and utility in clinical trials and longitudinal cohorts.
References
Footnotes
SJP and MØ contributed equally.
Contributors MØ, SJP and IJS designed the trial. JMM obtained MR images. IJS, BJ, ORM and SJP collected clinical data. SJP and SK were the primary MRI readers that read all spinal MR images according to the CANDEN and the SPARCC method. WPM read all spinal MR images according to the SPARCC method. SK performed data management and statistical analyses and drafted the manuscript. All authors critically revised the manuscript.
Funding The study was based on data from an investigator-initiated study supported by AbbVie. AbbVie had no role in the study design or in the collection, analysis or interpretation of the data, the writing of the manuscript or the decision to submit the manuscript for publication, and publication of this article was not contingent on approval by AbbVie. SK has received a PhD fellowship grant from The Danish Rheumatism Association (A3866).
Competing interests None declared.
Patient consent All patients gave written informed consent.
Ethics approval The study was approved by the Danish Health Authority and the Regional Ethics Committees in the Capital Region of Denmark, ethics approval number: H1-2013-118.
Provenance and peer review Not commissioned; externally peer reviewed.
Data sharing statement Data may be shared after agreement with MØ.