Abstract
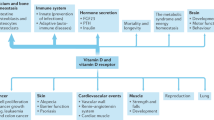
Vitamin D is a dietary vitamin that can also be synthesized in adequate amounts from cholesterol in most mammals exposed to sunlight. Vitamin D has classical roles in calcium and phosphate metabolism, and thus the skeleton; however, this molecule also has nonclassical effects that might influence the function of the immune, cardiovascular and endocrine systems. Vitamin D deficiency, due to insufficient sunlight exposure, dietary uptake and/or abnormalities in its metabolism, has been associated with rheumatic diseases, and both the classical and nonclassical effects of vitamin D might be of relevance to patients with rheumatic disease. However, conclusive data from intervention trials demonstrating the relationship between vitamin D levels and pathogenetic processes separate from classical effects of this molecule are lacking. Furthermore, the majority of studies linking vitamin D to health outcomes, harmful or beneficial, are observational in nature, linking clinical events to vitamin D exposure or serum levels of vitamin D metabolites. Evidence from high quality, prospective, double-blind, placebo-controlled, randomized trials should be obtained before vitamin D supplementation is recommended in the treatment of the many rheumatic conditions in which deficiency of this compound has been implicated. Herein, we review the evidence for vitamin D supplementation in the management of patients with rheumatic diseases.
Key Points
-
Vitamin D has classical roles in calcium and phosphate metabolism that influence the skeleton, as well as putative nonclassical effects that affect the cardiovascular, endocrine and immune systems
-
Vitamin D deficiency has been associated with a number of diseases, among them rheumatic diseases, including osteoporosis, osteoarthritis, rheumatoid arthritis and connective tissue diseases
-
Vitamin D given with calcium supplements modestly reduces the risk of osteoporotic fractures
-
Few intervention trials with vitamin D have been designed to address nonskeletal outcomes
-
Insufficient data are available to inform public health recommendations regarding vitamin D supplementation in rheumatic diseases other than osteoporosis
-
High quality prospective, double-blind, placebo-controlled randomized trials of vitamin D supplementation are needed
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Perwad, F. & Portale, A. A. Vitamin D metabolism in the kidney: regulation by phosphorus and fibroblast growth factor 23. Mol. Cell. Endocrinol. 347, 17–24 (2011).
St-Arnaud, R. Targeted inactivation of vitamin D hydroxylases in mice. Bone 25, 127–129 (1999).
Holick, M. F. et al. 1,24,25-trihydroxyvitamin D3. A metabolite of vitamin D3 effective on intestine. J. Biol. Chem. 248, 6691–6696 (1973).
Boyle, I. T., Omdahl, J. L., Gray, R. W. & DeLuca, H. F. The biological activity and metabolism of 24,25-dihydroxyvitamin D3 . J. Biol. Chem. 248, 4174–4180 (1973).
Ross, A. C., Taylor, C. L., Yaktine, A. L. & Del, H. B. IOM Dietary Reference Intakes for Calcium and Vitamin D. (The National Academies Press: Washington DC, 2011).
Adams, J. S. & Hewison, M. Extrarenal expression of the 25-hydroxyvitamin D-1-hydroxylase. Arch. Biochem. Biophys. 523, 95–102 (2012).
Gauzzi, M. C. et al. Suppressive effect of 1α,25-dihydroxyvitamin D3 on type I IFN-mediated monocyte differentiation into dendritic cells: impairment of functional activities and chemotaxis. J. Immunol. 174, 270–276 (2005).
Wang, T. T. et al. Cutting edge: 1,25-dihydroxyvitamin D3 is a direct inducer of antimicrobial peptide gene expression. J. Immunol. 173, 2909–2912 (2004).
Peelen, E. et al. Effects of vitamin D on the peripheral adaptive immune system: a review. Autoimmun. Rev. 10, 733–743 (2011).
Harvey, N. C. & Cooper, C. Vitamin D: some perspective please. BMJ 345, e4695 (2012).
Mavroeidi, A. et al. Seasonal 25-hydroxyvitamin D changes in British postmenopausal women at 57 degrees N and 51 degrees N: a longitudinal study. J. Steroid Biochem. Mol. Biol. 121, 459–461 (2010).
Thuesen, B. et al. Determinants of vitamin D status in a general population of Danish adults. Bone 50, 605–610 (2012).
Arabi, A., El Rassi, R. & El-Hajj Fuleihan, G. Hypovitaminosis D in developing countries—prevalence, risk factors and outcomes. Nat. Rev. Endocrinol. 6, 550–561 (2010).
Wallace, A. M., Gibson, S., De la Hunty, A., Lamberg-Allardt, C. & Ashwell, M. Measurement of 25-hydroxyvitamin D in the clinical laboratory: current procedures, performance characteristics and limitations. Steroids 75, 477–488 (2010).
Bolland, M. J., Grey, A. & Reid, I. R. Vitamin D and falls. Time for a moratorium on vitamin D meta-analyses? BMJ 339, b4394 (2009).
Tang, B. M., Eslick, G. D., Nowson, C., Smith, C. & Bensoussan, A. Use of calcium or calcium in combination with vitamin D supplementation to prevent fractures and bone loss in people aged 50 years and older: a meta-analysis. Lancet 370, 657–666 (2007).
Abrahamsen, B. et al. Patient level pooled analysis of 68,500 patients from seven major vitamin D fracture trials in US and Europe. BMJ 340, b5463 (2010).
Bischoff-Ferrari, H. A. et al. A pooled analysis of vitamin D dose requirements for fracture prevention. N. Engl. J. Med. 367, 40–49 (2012).
Rosen, C. J. et al. IOM committee members respond to endocrine society vitamin D guideline. J. Clin. Endocrinol. Metab. 97, 1146–1152 (2012).
Holick, M. F. et al. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 96, 1911–1930 (2011).
Heikinheimo, R. J. et al. Annual injection of vitamin D and fractures of aged bones. Calcif. Tissue Int. 51, 105–110 (1992).
Larsen, E. R., Mosekilde, L. & Foldspang, A. Vitamin D and calcium supplementation prevents osteoporotic fractures in elderly community dwelling residents: a pragmatic population-based 3-year intervention study. J. Bone Miner. Res. 19, 370–378 (2004).
Cummings, S. R. et al. Endogenous hormones and the risk of hip and vertebral fractures among older women. Study of Osteoporotic Fractures Research Group. N. Engl. J. Med. 339, 733–738 (1998).
[No authors listed] Recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis. American College of Rheumatology Task Force on Osteoporosis guidelines. Arthritis Rheum. 39, 1791–1801 (1996).
[No authors listed] ACR Recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis: 2001 update. American College of Rheumatology Ad Hoc Committee on Glucocorticoid-Induced Osteoporosis. Arthritis Rheum. 44, 1496–1503 (2001).
Grossman, J. M. et al. American College of Rheumatology 2010 recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Care Res. 62, 1515–1526 (2010).
Sambrook, P. et al. Prevention of corticosteroid osteoporosis. N. Engl. J. Med. 328, 1747–1752 (1993).
Buckley, L. M., Leib, E. S., Cartularo, K. S., Vacek, P. M. & Cooper, S. M. Calcium and vitamin D3 supplementation prevents bone loss in the spine secondary to low-dose corticosteroids in patients with rheumatoid arthritis. A randomized, double-blind, placebo-controlled trial. Ann. Intern. Med. 125, 961–968 (1996).
Adachi, J. D. et al. Vitamin D and calcium in the prevention of corticosteroid induced osteoporosis: a 3 year followup. J. Rheumatol. 23, 995–1000 (1996).
De Nijs, R. N. et al. Alendronate or alfacalcidol in glucocorticoid-induced osteoporosis. N. Engl. J. Med. 355, 675–684 (2006).
Silverman, S. Bazedoxifene in reducing new vertebral fracture risk in postmenopausal women with osteoporosis: results from a 3-year, randomized, placebo and active controlled trial. J. Bone Miner. Res. 23, 1923–1934 (2008).
Black, D. M. et al. Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Lancet 348, 1535–1541 (1996).
McCloskey, E. V. et al. Clodronate reduces the incidence of fractures in community-dwelling elderly women unselected for osteoporosis: results of a double-blind, placebo-controlled randomized study. J. Bone Miner. Res. 22, 135–141 (2007).
Bourke, S. et al. The impact of dietary calcium intake and vitamin D status on the effects of zoledronate. Osteoporos. Int. 24, 349–354 (2012).
Srikuea, R., Zhang, X., Park-Sarge, O.-K. & Esser, K. A. VDR and CYP27B1 are expressed in C2C12 cells and regenerating skeletal muscle: potential role in suppression of myoblast proliferation. Am. J. Physiol. Cell Physiol. 303, C396–C405 (2012).
Wang, Y. & DeLuca, H. F. Is the vitamin D receptor found in muscle? Endocrinology 152, 354–363 (2011).
Wang, Y., Zhu, J. & DeLuca, H. F. Where is the vitamin D receptor? Arch. Biochem. Biophys. 523, 123–133 (2012).
Gerdhem, P., Ringsberg, K. A., Obrant, K. J. & Akesson, K. Association between 25-hydroxyvitamin D levels, physical activity, muscle strength and fractures in the prospective population-based OPRA Study of Elderly Women. Osteoporos. Int. 16, 1425–1431 (2005).
Rosen, C. J. Vitamin D and falls—are intermittent, high doses better? Nat. Rev. Endocrinol. 7, 695–696 (2011).
Glendenning, P. et al. Effects of three monthly oral 150,000 IU cholecalciferol supplementation on falls, mobility and muscle strength in older postmenopausal women: a randomised controlled trial. J. Bone Miner. Res. 27, 170–176 (2011).
Sanders, K. M. et al. Annual high-dose oral vitamin D and falls and fractures in older women: a randomized controlled trial. JAMA 303, 1815–1822 (2010).
Murad, M. H. et al. Clinical review: The effect of vitamin D on falls: a systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 96, 2997–3006 (2011).
Thudi, A., Yin, S., Wandstrat, A. E., Li, Q. Z. & Olsen, N. J. Vitamin D levels and disease status in Texas patients with systemic lupus erythematosus. Am. J. Med. Sci. 335, 99–104 (2008).
Müller, K. et al. Vitamin D3 metabolism in patients with rheumatic diseases: low serum levels of 25-hydroxyvitamin D3 in patients with systemic lupus erythematosus. Clin. Rheumatol. 14, 397–400 (1995).
Fragoso, T. S. et al. 25-Hydroxyivitamin D3 levels in patients with systemic lupus erythematosus and its association with clinical parameters and laboratory tests. Rev. Bras. Reumatol. 52, 60–65 (2012).
Hamza, R. T., Awwad, K. S., Ali, M. K. & Hamed, A. I. Reduced serum concentrations of 25-hydroxy vitamin D in Egyptian patients with systemic lupus erythematosus: relation to disease activity. Med. Sci. Monit. 17, CR711–CR718 (2011).
Stockton, K. A., Kandiah, D. A., Paratz, J. D. & Bennell, K. L. Fatigue, muscle strength and vitamin D status in women with systemic lupus erythematosus compared with healthy controls. Lupus 21, 271–278 (2012).
Souto, M. et al. Vitamin D insufficiency in Brazilian patients with SLE: prevalence, associated factors, and relationship with activity. Lupus 20, 1019–1026 (2011).
Lee, Y. H., Bae, S. C., Choi, S. J., Ji, J. D. & Song, G. G. Associations between vitamin D receptor polymorphisms and susceptibility to rheumatoid arthritis and systemic lupus erythematosus: a meta-analysis. Mol. Biol. Rep. 38, 3643–3651 (2011).
Mok, C. C. et al. Vitamin D deficiency as marker for disease activity and damage in systemic lupus erythematosus: a comparison with anti-dsDNA and anti-C1q. Lupus 21, 36–42 (2012).
Szodoray, P. et al. The immunopathological role of vitamin D in patients with SLE: data from a single centre registry in Hungary. Scand. J. Rheumatol. 40, 122–126 (2011).
Birmingham, D. J. et al. Evidence that abnormally large seasonal declines in vitamin D status may trigger SLE flare in non-African Americans. Lupus 21, 855–864 (2012).
Reid, D. et al. The relation between acute changes in the systemic inflammatory response and plasma 25-hydroxyvitamin D concentrations after elective knee arthroplasty. Am. J. Clin. Nutr. 93, 1006–1011 (2011).
Gama, R. et al. Hypovitaminosis D and disease: consequence rather than cause? BMJ 345, e5706 (2012).
Heine, G., Lahl, A., Müller, C. & Worm, M. Vitamin D deficiency in patients with cutaneous lupus erythematosus is prevalent throughout the year. Br. J. Dermatol. 163, 863–865 (2010).
Cutillas-Marco, E., Morales-Suárez-Varela, M., Marquina-Vila, A. & Grant, W. Serum 25-hydroxyvitamin D levels in patients with cutaneous lupus erythematosus in a Mediterranean region. Lupus 19, 810–814 (2010).
Cusack, C. et al. Photoprotective behaviour and sunscreen use: impact on vitamin D levels in cutaneous lupus erythematosus. Photodermatol. Photoimmunol. Photomed. 24, 260–267 (2008).
Costenbader, K. H., Feskanich, D., Holmes, M., Karlson, E. W. & Benito-Garcia, E. Vitamin D intake and risks of systemic lupus erythematosus and rheumatoid arthritis in women. Ann. Rheum. Dis. 67, 530–535 (2008).
Hacker-Thompson, A., Schloetter, M. & Sellmeyer, D. E. Validation of a dietary vitamin D questionnaire using multiple diet records and the block 98 health habits and history questionnaire in healthy postmenopausal women in northern California. J. Acad. Nutr. Diet. 112, 419–423 (2012).
Ruiz-Irastorza, G., Gordo, S., Olivares, N., Egurbide, M. V. & Aguirre, C. Changes in vitamin D levels in patients with systemic lupus erythematosus: effects on fatigue, disease activity, and damage. Arthritis Care Res. 62, 1160–1165 (2010).
Aghdassi, E. et al. The use of micronutrient supplements is not associated with better quality of life and disease activity in Canadian patients with systemic lupus erythematosus. J. Rheumatol. 37, 87–90 (2010).
Azali, P. et al. Low serum levels of vitamin D in idiopathic inflammatory myopathies. Ann. Rheum. Dis. 72, 512–516 (2013).
Gambichler, T., Chrobok, I., Höxtermann, S. & Kreuter, A. Significantly decreased serum 25-hydroxyvitamin d levels in a large German systemic sclerosis cohort. J. Rheumatol. 38, 2492–2493 (2011).
Rios Fernández, R., Fernández Roldán, C., Callejas Rubio, J. L. & Ortego Centeno, N. Vitamin D deficiency in a cohort of patients with systemic scleroderma from the south of Spain. J. Rheumatol. 37, 1355; author reply 1356 (2010).
Vacca, A. et al. Vitamin D deficiency and insufficiency in 2 independent cohorts of patients with systemic sclerosis. J. Rheumatol. 36, 1924–1929 (2009).
Caramaschi, P. et al. Very low levels of vitamin D in systemic sclerosis patients. Clin. Rheumatol. 29, 1419–1425 (2010).
Hulshof, M. M. et al. Double-blind, placebo-controlled study of oral calcitriol for the treatment of localized and systemic scleroderma. J. Am. Acad. Dermatol. 43, 1017–1023 (2000).
Cutolo, M. Rheumatoid arthritis: circadian and circannual rhythms in RA. Nat. Rev. Rheumatol. 7, 500–502 (2011).
Cutolo, M. et al. Circannual vitamin D serum levels and disease activity in rheumatoid arthritis: Northern versus Southern Europe. Clin. Exp. Rheumatol. 24, 702–704 (2006).
Rossini, M. et al. Relationship of focal erosions, bone mineral density, and parathyroid hormone in rheumatoid arthritis. J. Rheumatol. 38, 997–1002 (2011).
Rossini, M. et al. Vitamin D deficiency in rheumatoid arthritis: prevalence, determinants and associations with disease activity and disability. Arthritis Res. Ther. 12, R216 (2010).
Kerr, G. S. et al. Prevalence of vitamin D insufficiency/deficiency in rheumatoid arthritis and associations with disease severity and activity. J. Rheumatol. 38, 53–59 (2011).
Haque, U. J. & Bartlett, S. J. Relationships among vitamin D, disease activity, pain and disability in rheumatoid arthritis. Clin. Exp. Rheumatol. 28, 745–747 (2010).
Turhanoğlu, A. D. et al. The relationship between vitamin D and disease activity and functional health status in rheumatoid arthritis. Rheumatol. Int. 31, 911–914 (2011).
Feser, M. et al. Plasma 25,OH vitamin D concentrations are not associated with rheumatoid arthritis (RA)-related autoantibodies in individuals at elevated risk for RA. J. Rheumatol. 36, 943–946 (2009).
Craig, S. M. et al. Vitamin D status and its associations with disease activity and severity in African Americans with recent-onset rheumatoid arthritis. J. Rheumatol. 37, 275–281 (2010).
Merlino, L. A. et al. Vitamin D intake is inversely associated with rheumatoid arthritis: results from the Iowa Women's Health Study. Arthritis Rheum. 50, 72–77 (2004).
Andjelkovic, Z. et al. Disease modifying and immunomodulatory effects of high dose 1α(OH)D3 in rheumatoid arthritis patients. Clin. Exp. Rheumatol. 17, 453–456 (1999).
Uitterlinden, A. G. et al. Vitamin D receptor genotype is associated with radiographic osteoarthritis at the knee. J. Clin. Invest. 100, 259–263 (1997).
Huang, J., Ushiyama, T., Inoue, K., Kawasaki, T. & Hukuda, S. Vitamin D receptor gene polymorphisms and osteoarthritis of the hand, hip, and knee: a case–control study in Japan. Rheumatology (Oxford) 39, 79–84 (2000).
Videman, T. et al. The relative roles of intragenic polymorphisms of the vitamin D receptor gene in lumbar spine degeneration and bone density. Spine (Phila. Pa. 1976) 26, E7–E12 (2001).
Solovieva, S. et al. Vitamin D receptor gene polymorphisms and susceptibility of hand osteoarthritis in Finnish women. Arthritis Res. Ther. 8, R20 (2006).
arcOGEN Constorium and arcOGEN Collaborators. Identification of new susceptibility loci for osteoarthritis (arcOGEN): a genome-wide association study. Lancet 380, 815–823 (2012).
Chaganti, R. K. et al. Association of 25-hydroxyvitamin D with prevalent osteoarthritis of the hip in elderly men: the osteoporotic fractures in men study. Arthritis Rheum. 62, 511–514 (2010).
Lane, N. E. et al. Serum vitamin D levels and incident changes of radiographic hip osteoarthritis: a longitudinal study. Study of Osteoporotic Fractures Research Group. Arthritis Rheum. 42, 854–860 (1999).
Ding, C. et al. Serum levels of vitamin D, sunlight exposure, and knee cartilage loss in older adults: the Tasmanian older adult cohort study. Arthritis Rheum. 60, 1381–1389 (2009).
Bergink, A. P. et al. Vitamin D status, bone mineral density, and the development of radiographic osteoarthritis of the knee: the Rotterdam Study. J. Clin. Rheumatol. 15, 230–237 (2009).
Felson, D. T. et al. Low levels of vitamin D and worsening of knee osteoarthritis: results of two longitudinal studies. Arthritis Rheum. 56, 129–136 (2007).
Muraki, S. et al. Association of vitamin D status with knee pain and radiographic knee osteoarthritis. Osteoarthritis Cartilage 19, 1301–1306 (2011).
Lange, U., Jung, O., Teichmann, J. & Neeck, G. Relationship between disease activity and serum levels of vitamin D metabolites and parathyroid hormone in ankylosing spondylitis. Osteoporos. Int. 12, 1031–1035 (2001).
Jung, K. H. et al. Associations of vitamin D binding protein gene polymorphisms with the development of peripheral arthritis and uveitis in ankylosing spondylitis. J. Rheumatol. 38, 2224–2229 (2011).
Fischer, R. et al. Discovery of candidate serum proteomic and metabolomic biomarkers in ankylosing spondylitis. Mol. Cell. Proteomics 11, M111.013904 (2012).
Touma, Z. et al. Seasonal variation in vitamin D levels in psoriatic arthritis patients from different latitudes and its association with clinical outcomes. Arthritis Care Res. 63, 1440–1447 (2011).
Huckins, D., Felson, D. T. & Holick, M. Treatment of psoriatic arthritis with oral 1,25-dihydroxyvitamin D3: a pilot study. Arthritis Rheum. 33, 1723–1727 (1990).
Heidari, B., Shirvani, J. S., Firouzjahi, A., Heidari, P. & Hajian-Tilaki, K. O. Association between nonspecific skeletal pain and vitamin D deficiency. Int. J. Rheum. Dis. 13, 340–346 (2010).
Bhatty, S. A. et al. Vitamin D deficiency in fibromyalgia. J. Pak. Med. Assoc. 60, 949–951 (2010).
Macfarlane, G. J. et al. An excess of widespread pain among South Asians: are low levels of vitamin D implicated? Ann. Rheum. Dis. 64, 1217–1219 (2005).
McBeth, J. et al. Musculoskeletal pain is associated with very low levels of vitamin D in men: results from the European Male Ageing Study. Ann. Rheum. Dis. 69, 1448–1452 (2010).
Straube, S., Derry, S., Moore, R. A. & McQuay, H. J. Vitamin D for the treatment of chronic painful conditions in adults. Cochrane Database of Systemic Reviews, Issue 1. Art. No.: CD007771 http://dx.doi.org/10.1002/14651858.CD007771.pub2.
Brohult, J. & Jonson, B. Effects of large doses of calciferol on patients with rheumatoid arthritis. A double-blind clinical trial. Scand. J. Rheumatol. 2, 173–176 (1973).
Di Munno, O. et al. Prevention of glucocorticoid-induced osteopenia: effect of oral 25-hydroxyvitamin D and calcium. Clin. Rheumatol. 8, 202–207 (1989).
Warner, A. E. & Arnspiger, S. A. Diffuse musculoskeletal pain is not associated with low vitamin D levels or improved by treatment with vitamin D. J. Clin. Rheumatol. 14, 12–16 (2008).
Yamauchi, Y. et al. A double blind trial of alfacalcidol on patients with rheumatoid arthritis (RA) [Japanese]. Ryumachi. 29, 11–24 (1989).
Richart, T., Li, Y. & Staessen, J. A. Renal versus extrarenal activation of vitamin D in relation to atherosclerosis, arterial stiffening, and hypertension. Am. J. Hypertens. 20, 1007–1015 (2007).
Michos, E. D. et al. 25-hydroxyvitamin D deficiency is associated with fatal stroke among whites but not blacks: The NHANES-III linked mortality files. Nutrition 28, 367–371 (2012).
Thomas, G. N. et al. Vitamin D levels predict all-cause and cardiovascular disease mortality in subjects with the metabolic syndrome: the Ludwigshafen Risk and Cardiovascular Health (LURIC) Study. Diabetes Care 35, 1158–1164 (2012).
Bolland, M. J. et al. Vitamin D insufficiency and health outcomes over 5 y in older women. Am. J. Clin. Nutr. 91, 82–89 (2010).
Wasson, L. T. et al. Is vitamin D deficiency a risk factor for ischemic heart disease in patients with established cardiovascular disease? 10-year follow-up of the Nova Scotia Health Survey. Int. J. Cardiol. 148, 387–389 (2011).
Liu, L. C. Y. et al. Vitamin D status and outcomes in heart failure patients. Eur. J. Heart Fail. 13, 619–625 (2011).
Hsia, J. et al. Calcium/vitamin D supplementation and cardiovascular events. Circulation 115, 846–854 (2007).
Bolland, M. J. et al. Effect of calcium supplements on risk of myocardial infarction and cardiovascular events: meta-analysis. BMJ 341, c3691 (2010).
Prince, R., Zhu, K. & Lewis, J. R. Evidence of harm is unconvincing. BMJ 342, d3541 (2011).
Abrahamsen, B. & Sahota, O. Do calcium plus vitamin D supplements increase cardiovascular risk? BMJ 342, d2080 (2011).
Aspray, T. J. & Francis, R. M. Calcium and vitamin D supplementation and cardiovascular disease: quo vadis? Maturitas 69, 285–286 (2011).
Jackson, R. D. et al. Calcium plus vitamin D supplementation and the risk of fractures. N. Engl. J. Med. 354, 669–683 (2006).
Freedman, D. M., Looker, A. C., Abnet, C. C., Linet, M. S. & Graubard, B. I. Serum 25-hydroxyvitamin D and cancer mortality in the NHANES III study (1988–2006). Cancer Res. 70, 8587–8597 (2010).
Chung, M., Lee, J., Terasawa, T., Lau, J. & Trikalinos, T. A. Vitamin D with or without calcium supplementation for prevention of cancer and fractures: an updated meta-analysis for the U.S. Preventive Services Task Force. Ann. Intern. Med. 155, 827–838 (2011).
Morimoto, T. et al. Epidemiology of sarcoidosis in Japan. Eur. Respir. J. 31, 372–379 (2008).
Sharma, O. P. Hypercalcemia in granulomatous disorders: a clinical review. Curr. Opin. Pulm. Med. 6, 442–447 (2000).
Burke, R. R., Rybicki, B. A. & Rao, D. S. Calcium and vitamin D in sarcoidosis: how to assess and manage. Semin. Respir. Crit. Care Med. 31, 474–484 (2010).
Kavathia, D., Buckley, J. D., Rao, D., Rybicki, B. & Burke, R. Elevated 1,25-dihydroxyvitamin D levels are associated with protracted treatment in sarcoidosis. Respir. Med. 104, 564–570 (2010).
Bolland, M. J., Grey, A., Avenell, A., Gamble, G. D. & Reid, I. R. Calcium supplements with or without vitamin D and risk of cardiovascular events: reanalysis of the Women's Health Initiative limited access dataset and meta-analysis. BMJ 342, d2040 (2011).
Author information
Authors and Affiliations
Contributions
Both authors contributed equally to all stages of the preparation of this manuscript for publication.
Corresponding author
Ethics declarations
Competing interests
B. Abrahamsen declares that he has acted as a consultant for Amgen and Takeda/Nycomed, has received research support from Amgen, Novartis and NPS Pharmaceuticals, and has received speakers bureau (honoraria) from Amgen, Eli Lilly, Merck and Nycomed. N. C. Harvey declares that he has received research support from Servier, and has received speakers bureau (honoraria) from the Alliance for Better Bone Health, Amgen, Lilly, Nycomed, Proctor and Gamble, Sanofi-Aventis, Servier and Shire.
Supplementary information
Supplementary Table 1
Studies of vitamin D and fractures, falls and BMD (DOC 209 kb)
Supplementary Table 2
Studies of vitamin D in SLE or CLE (DOC 199 kb)
Supplementary Table 3
Studies of vitamin D in SSc and myopathy (DOC 148 kb)
Supplementary Table 4
Studies of vitamin D in RA (DOC 198 kb)
Supplementary Table 5
Studies of vitamin D in OA (DOC 185 kb)
Supplementary Table 6
Studies of vitamin D in seronegative diseases and chronic pain syndromes (DOC 199 kb)
Rights and permissions
About this article
Cite this article
Abrahamsen, B., Harvey, N. The role of vitamin D supplementation in patients with rheumatic diseases. Nat Rev Rheumatol 9, 411–422 (2013). https://doi.org/10.1038/nrrheum.2013.71
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2013.71
This article is cited by
-
Conjunctival impression cytology and tear-film changes in cases with vitamin D deficiency
International Ophthalmology (2020)
-
Screening for vitamin D deficiency in a tropical area: results of a sun exposure questionnaire
BMC Endocrine Disorders (2018)
-
Vitamin D Deficiency and Rheumatoid Arthritis
Clinical Reviews in Allergy & Immunology (2017)
-
The association of vitamin D deficiency with tear break-up time and Schirmer testing in non-Sjögren dry eye
Eye (2015)